TPS Device
Neurolith is a revolutionary non-invasive neuromodulation device that uses Transcranial Pulse Stimulation (TPS) to stimulate deep brain regions linked to cognitive function.

Neurolith is a leader in evidence-based therapy for Alzheimer’s, and combines CE-approved engineering, strong clinical validation, minimal side effects, and user-friendly delivery, making it a safe and effective solution for supporting brain health.
Explore the extensive clinical research underpinning neuromodulation techniques. A wealth of peer-reviewed scientific articles is easily accessible, with a continuous stream of new research papers emerging each year. These publications consistently reinforce the safety profiles of neuromodulation devices.
Our methodology relies on evidence-based practices designed to address precise cognitive functions linked to brain-affected diseases. Delve into the scientific literature on neuromodulation techniques with us.
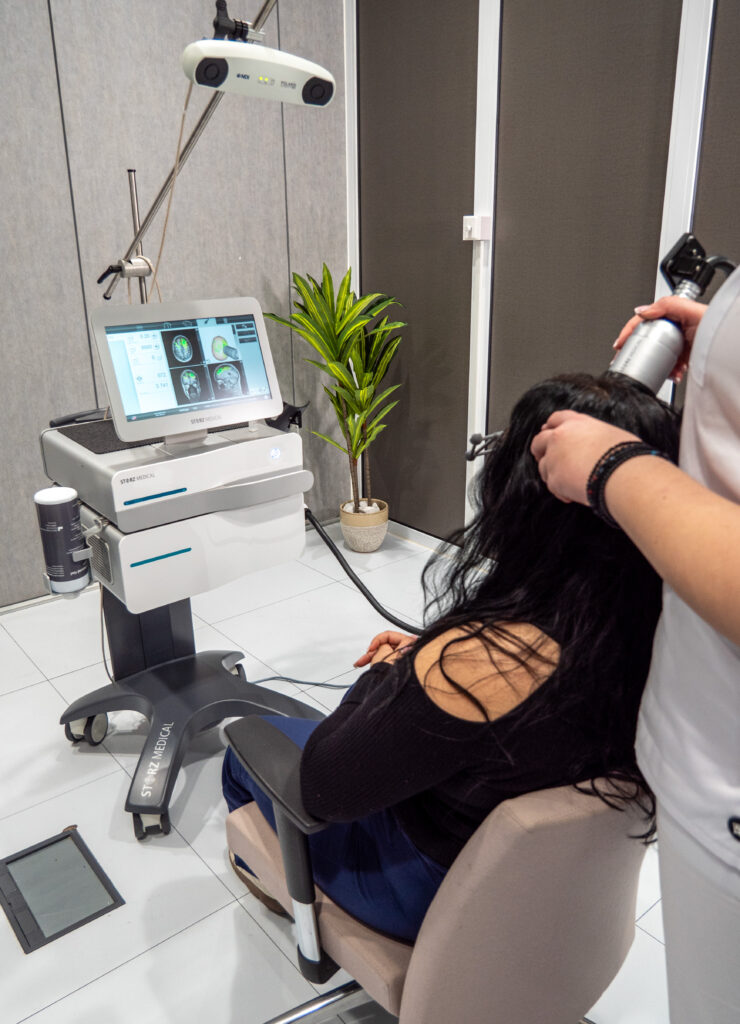
Neurolith was primarily designed for individuals with mild to moderate Alzheimer’s disease and related cognitive decline, and clinical studies have shown that the device can help maintain or temporarily improve cognitive function, with average gains of approximately 10 points on the CERAD scale. Users have also demonstrated enhancements in memory, language abilities, orientation, and overall quality of life.
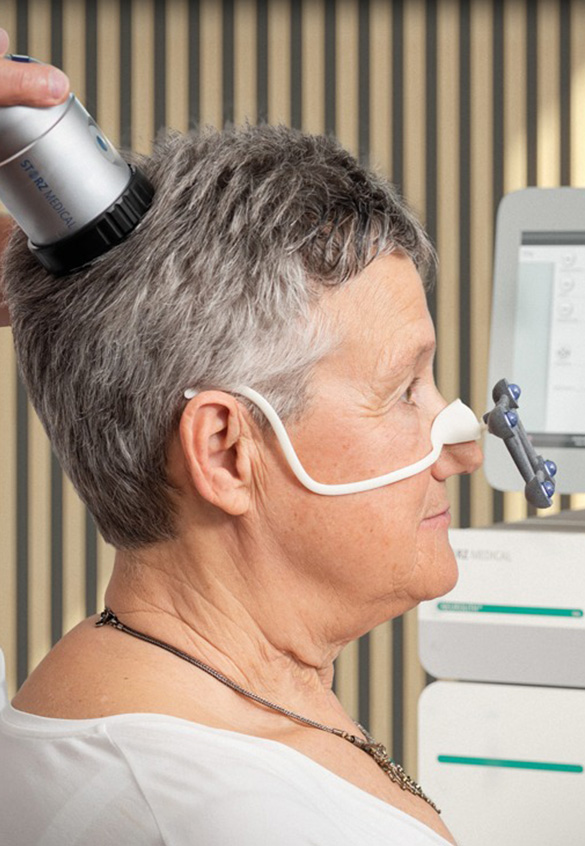
Neurolith provides Transcranial Pulse Stimulation (TPS), delivering focused acoustic pulses through the skull to reach deep brain regions up to approximately 8 cm which are affected by Alzheimer’s disease and Dementia. This non-invasive brain therapy enhances cerebral blood flow, supports nerve regeneration, and increases essential growth factors such as VEGF and BDNF, helping to improve cognitive function and overall brain health.
A growing body of peer-reviewed research supports Neurolith’s efficacy and durability, while multiple studies show long-term effects, reduced cortical atrophy, and even mood improvements.
Some studies include:
For more Scientific Literature visit https://sozobraincenter.com/scientific-research/
Neurolith is CEmarked since December 2018, and validated and approved for Alzheimer’s therapy. Clinical routines utilize navigated systems like BodyTrack™ for precision and safety. Emergency-authority approvals are underway (e.g., FDA investigational use via distributor Soterix Medical in the US).
Neurolith is non-invasive with an outpatient-friendly design. There is no need for hair shaving or sedation and patients remain alert and comfortable.
Some patients may experience minor side effects like mild headaches, fatigue, or temporary redness; however, these side effects are rare and transient.
Neurolith is not suitable for individuals with:
Coagulation disorders or thrombosis
Brain tumors in the treatment area
Recent high-dose corticosteroid therapy (within 6 weeks)
Metal implants in the skull or non-TPS-compatible pacemakers / devices
Pregnancy
Contact our medical team, book your appointment at SOZΩ Brain Center, and start your journey with TPS